|
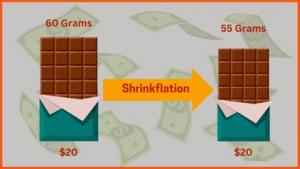
| ▲ A pharmacy in Seoul posted a notice saying that the self-diagnosis kit was out of stock. (Sources: YTN) |
On the 13th, the government announced a policy to ban online sales of COVID-19 self-inspection kits and restrict the amount to five per person based on one place of purchase when purchasing offline. The quarantine authorities seem to have put forward this policy to prevent the shortage of self-diagnosis kits and hoarding for profit acquisition due to the prolonged pandemic. Accordingly, the Ministry of Food and Drug Safety announced that it will supply 30 million self-diagnosis kits that can be purchased individually from pharmacies and convenience stores in accordance with measures to improve the distribution of self-inspection kits.
However, the public responded that they were not happy with the turn of events regarding the quarantine system. This is because the government stresses day after day that the amount of kits is sufficient, however this is not the case with the public. There was a shortage of supplies throughout the store, with most of them being sold out at the same time as they were received. There was even a fraud case that exploited this confusion. On the 11th, an official letter was faxed to pharmacies in Jongno-gu, Seoul, stating that they would provide a special self-diagnosis kit. The official letter contained the name of a specific manufacturer of the self-diagnosis kit, and the unit price was about 2,000 won cheaper than the current market price. However, the diagnostic kit written in the official document was soon found to be a product that was not approved for sale in Korea. In response, the Korean Pharmaceutical Association strongly suspected it as a fraudulent act that sent fake official letters to pharmacies followed by demanding a method of receipt after paying in advance.
In addition, even if it is not a fraud aimed at a seller, online violations are constantly occurring. Recently, when the government turned to a self-quarantine system in response to COVID-19, self-diagnosis kits were scarce. Despite the ban on online sales, there are frequent cases of people who hoard self-diagnosis kits using second hand trading platforms to sell goods. For example, it was deleted about an hour after a post appeared that said it would sell 120,000 won for 24 kits on the platform "Beon-jang" and 150,000 won for 10 kits on the platform "Dang-geun Market." Despite a series of censorship on these platforms, this behavior continues. Until now, the Ministry of Food and Drug Safety has tolerated one-time small amount of personal used transactions. According to the authoritative interpretation of the Ministry of Food and Drug Safety, one-off private transactions within the normal price range do not constitute commercial activities for profit. However, all online sales will be banned until the 5th of next month, so even small transactions that fall within the existing allowable range are subject to sanctions. Regarding this phenomenon, an official from the Ministry of Food and Drug Safety said, "We are continuously cracking down on transactions for profit, such as making profits beyond the allowable range, and in such cases, we are asking the police to investigate and seek criminal punishment."
The shortage of kits is also emerging as a social problem, but controversy is also brewing over the accuracy of self-inspection kits. According to the Central Disaster and Safety Countermeasure Headquarters, PCR tests at screening clinics are currently limited to those in close contact with confirmed patients, aged 60 or older, doctors requesting tests, vulnerable facility workers, and patients who have received positive results for quick antigen tests and emergency screening tests. In other cases, even if they have symptoms, they should do a self-examination kit at a screening clinic or get a quick antigen test from experts at a respiratory clinic first. However, it is known that the Korean Society of Diagnostic Tests analyzed the sensitivity of rapid antigen testing (self-test kit) at up to 41.5%, sparking controversy over the accuracy of the kit. Sensitivity refers to the probability of accurately confirming the positive test of a positive patient. Compared to the fact that the sensitivity of PCR tests is close to 99%. It is true that the accuracy is quite low. As self-examination kits are used in the first diagnosis process, people are increasingly worried that they may increase the risk in their daily lives because they cannot accurately determine whether they are infected.
Prime Minister Kim Bu-gyeom said, "I am sorry for the over 90,000 COVID-19 confirmed cases," stressing, "There is enough supply of inspection kits in February and March, so there is no need to buy a large number of inspection kits that are not needed immediately." With many people living in confusion due to changes in the quarantine system, it is time for the community to focus on overcoming the crisis together, aiming for ethical consumption, not for personal gain.
By Seo Ji-min, cub-reporter
Seo Ji-min, cub-reporter jmseo1215@naver.com
<저작권자 © The Campus Journal, 무단 전재 및 재배포 금지>
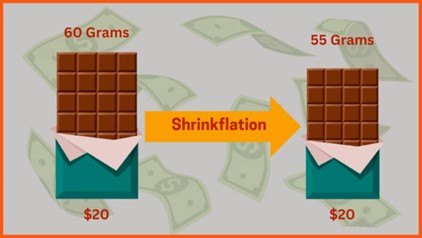 Shrinkflation, Consumer Deception
Shrinkflation, Consumer Deception